 |
|
|
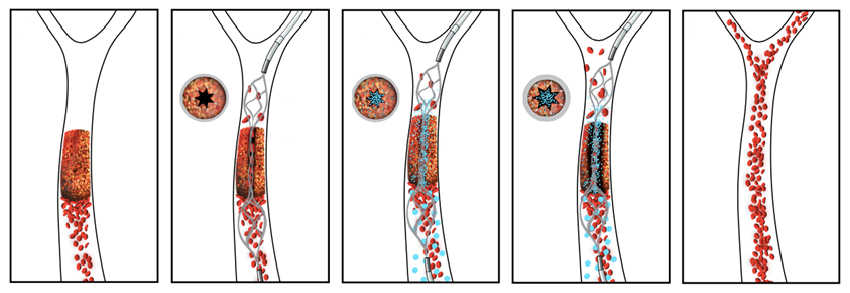
The new drug-device combination pairs an injectable clot-busting nanotherapeutic that targets blockages with an intra-arterial device that restores blood flow to obstructed vessels, as shown in this illustration. |
Scientists at the New England Center for Stroke Research at UMass Medical School and the Wyss Institute for Biologically Inspired Engineering at Harvard University are developing a drug-device combination for treating life-threatening blood clots in patients with stroke.
In a new study that will appear in the December issue of the journal Stroke, the team co-led by Ajay Wakhloo, MD, PhD, professor of radiology at UMMS, and Donald Ingber, MD, PhD, founding director of the Wyss Institute, describe their novel method to quickly dissolve clots that obstruct blood vessels in the brain. Their approach combines an injectable clot-busting nanotherapeutic that targets blockages with an intra-arterial device that restores blood flow to obstructed vessels.
The Wyss Institute nanotherapeutic is composed of an aggregate of biodegradable nanoparticles coated with a clot-busting drug called tissue plasminogen activator (tPA), which mimics the way blood platelets behave inside our own bodies. When blood vessels narrow, the force of blood flow increases at that location to produce a physical cue that causes platelets to stick to the vessel wall. Similarly, the nanotherapeutic reacts to fluid shear force, releasing tPA-coated nanoparticles in these narrowed regions where vessels are partially occluded, binding to the blood clot and dissolving it away.
But until now, the mechanically activated nanotherapeutic would not be effective in complete vascular blockages where there is no blood flow, as is the case for most stroke patients. The most effective treatment today for stroke is known as a “stent-retriever thrombectomy” procedure, originally described by Dr. Wakhloo and Matthew Gounis, PhD, associate professor of radiology. The procedure involves placing a small tube through the blockage, passing a closed stent through it and then opening the stent to physically pull the large blood clot out of the vessel.
“Even with the retriever thrombectomy procedure, not all clots can be removed with a successful outcome,” said Dr. Gounis, director of the New England Center for Stroke Research at UMMS. “Clot fragments can be dislodged, which can lead to microclots and tissue damage downstream in the brain circulatory system, and physical dragging of the stent through the vessel can potentially be damaging as well.”
Instead, the new advance describes using the stent not to drag out the clot, but to create a narrow channel restoring blood flow through an opening in the center of the vascular blockage. Doing so creates a high level of shear force generated by restored flow, activating the nanotherapeutic to release and target the clot-busting drug along the opened channel in the clot. After the blood clot is fully dissolved, the stent is re-sheathed and harmlessly removed from the vessel. If during the process any clot fragments break off and travel away through the circulatory system, the drug-coated nanoparticles will remain bound to them and continue to dissolve them locally wherever they go.
“What’s progressive about this approach is that the temporary opening of a tiny hole in the clot—using a stent device that is already commonly used clinically—results in a local rise in mechanical forces that activate the nanotherapeutic to deploy the clot-busting drug precisely where it can best do its job,” said Dr. Ingber, who is also the Judah Folkman Professor of Vascular Biology at Boston Children’s Hospital and Harvard Medical School and professor of bioengineering at the Harvard John A. Paulson School of Engineering and Applied Sciences.