Amyotrophic Lateral Sclerosis, SOD1-associated
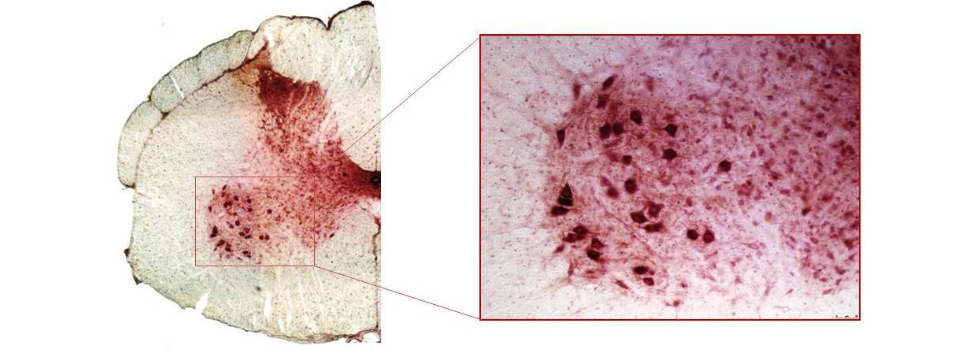
This image shows a monkey spinal cord, with a zoom on an area called the ventral horn. The dark red cells are motor neurons. The red shows presence of GFP, a protein that is not normally expressed by the animals. Its presence therefore demonstrates that we are able to transduce motor neurons with our gene therapy vector.
What is Amyotrophic lateral sclerosis?
Amyotrophic lateral sclerosis (ALS), also known as Charcot disease after the physician who first described it, and Lou Gehrig’s disease in the US after a famous baseball player who suffered from ALS, is a neurodegenerative disorder that primarily affects the motor neurons.
Clinically this leads to progressive muscle weakness, progressive paralysis and ultimately death within three to five years post-diagnosis. There is currently no cure for ALS. Despite the number of ongoing clinical trials, the only FDA-approved treatment to date is riluzole, which only increases patient survival by about 3 months. There is therefore an unmet medical need for this fatal disease, and several novel therapeutics are being actively investigated. One of the proposed strategies is gene therapy.
Our therapeutic strategy: silencing SOD1
Mutations in several genes have been associated with ALS. The first gene ever linked to ALS was the superoxide dismutase 1 (SOD1) gene. It was published in 1993 by Dr Robert H. Brown Jr, an internationally known researcher and physician, who joined UMass as Chair of Neurology in May 2008. In 2012, he teamed up with our lab to develop a gene therapy program for ALS. Combining his expertise in ALS and our lab's expertise in gene therapy and gene silencing have led to the design, validation, and preclinical development of a gene therapy product for SOD1-linked ALS.

Our therapeutic strategy is to silence SOD1 following CSF-directed delivery of an AAVrh.10 expressing an artificial miRNA targeting all forms of SOD1 (wild-type and mutant). To achieve this, we have designed a miRNA called miR-SOD1, and we have shown that it mediates a strong silencing of SOD1 in cell culture. We then proceeded to test this candidate in adult ALS mice (SOD1G93A) to mimic as well as possible the human disease. We also tested it in non-human primates, to assess the efficacy in a model that is larger and closer to human. In monkeys, we demonstrated that we are able to efficiently target the motor neurons (see image on top of this page).
NEW! Our latest publication
We have recently published the results of our preclinical studies for our miR-SOD1 candidate for the treatment of SOD1-linked ALS.
Learn more about the progress of this program here: we published in January 2016 the promising results of our preclinical studies in ALS mice and in monkeys. In this publication, we show that when treating adult ALS mice, we are able to delay both disease onset and death. In addition, the treated mice maintain their muscle strength, motor and respiratory function. We also demonstrate safety and efficacy in non-human primates.